Research In Action
Research In Action
Breadcrumb
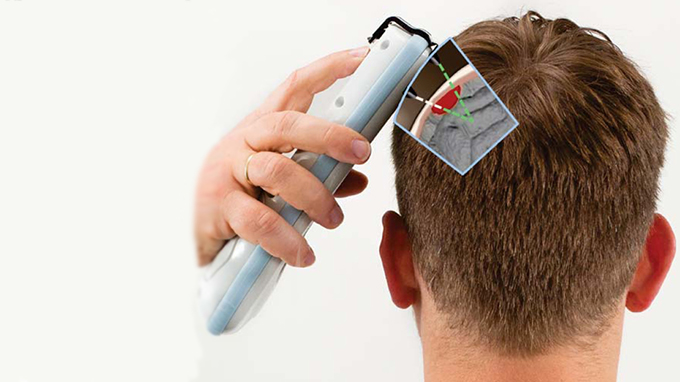
Colleagues at CHOP (including first author Dr. Matt Kirschen and CIRP Senior Fellow Dr. Sage Myers), Boston Children’s Hospital, Children’s Hospital Colorado and I recently published an article in the Western Journal of Emergency Medicine validating a handheld, near-infrared spectroscopy device (Infrascanner) for detecting intracranial hematomas (collections of blood) in children presenting to the emergency department (ED) with head injury.
The Infrascanner is a non-invasive, radiation sparing technology that measures the near-infrared light absorption of hemoglobin (a protein inside red blood cells) within the brain and can distinguish intracranial bleeds and the uninjured brain. While it has previously been validated among adult patients with head trauma, our study is the first to evaluate the most recent model of the Infrascanner in children using a multicenter approach.
What We Found
We analyzed data from 344 patients < 18 years old enrolled at the three hospitals’ EDs and compared the Infrascanner to head CT results to detect the hematomas. The sensitivity of the device (the ability to correctly identify all patients with a hematoma) was 81%. While this was high, it is not necessarily high enough to use the device by itself to screen for all hematomas.
Study Implications
We did, however, find that the negative predictive value (the probability that a patient did not have a hematoma with a negative test using the Infrascanner) was 99%. This shows that the device can be a useful adjunctive tool to existing prediction rules for head trauma that help predict whether or not a patient would need neuroimaging (such as a head CT or MRI) or may have a hematoma. It could be especially important for patients who fall into an equivocal risk category based on existing head trauma prediction rules.
Future studies can help determine optimal training paradigms and the clinical impact of the device, including further reduction of neuroimaging or prolonged observation in the ED.

